21 Rue de la Noue Bras De Fer
44 200 NANTES
FRANCE
i-SEP: a new-generation intraoperative cell salvage technology
Article published in the La Tribune
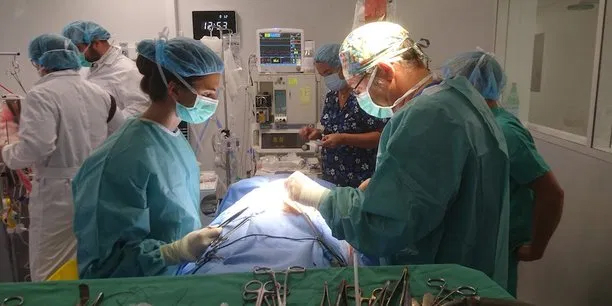
i-SEP has just been awarded a €3 million grant as part of PIA Investing-for-the-Future Program to finalize a new generation blood auto-transfusion device. The Nantes-based startup is preparing to launch clinical trials of this innovative device designed to make patient blood transfusions safer and save the healthcare system money.
In the form of a grant and a repayable advance, the Nantes-based startup i-SEP was recently allocated €3 million in aid as part of the PIA program in the PSPC category (Major Research and Development Projects supporting Competitiveness), which is open to any company with major research projects of between €5 and €50 million. The Pays de la Loire region is providing additional support to the tune of €500,000 via the European Regional Development Fund (ERDF). All of which will enable i-SEP to launch clinical trials in 2019, complete regulatory obligations and market its first products as early as 2020. Three years after its creation, i-SEP wants to move fast and, as part of its search for a new investor, is already planning another round of financing to raise €2 million.
“We expect this next round of funding to attract one or more investment funds specializing in Medtech into the capital. Combined with the existing funding from research programs, we expect to attain an investment capacity of between €6 million to €7 million to support the marketing of our auto-transfusion device within three years”, explains Sylvain Picot, Chairman of i-SEP, whose capital is currently jointly owned by the founders themselves, the Go Capital fund (early investors for €800,000 and €1.3 million) and business angels for €230,000.
A significant health matter for patients and the health care system
Since its creation, between research credits, subsidies, repayable advances and self-financing, the young company has put together a budget of €4 million to finance and develop the breakthrough technological solution invented by Françis Gadrat, an anesthetist and intensive care physician at Bordeaux University Hospital. In 2015, in partnership with Bertrand Chastenet, a consultant and manager in the pharmaceutical industry, and entrepreneur Sylvain Picot, he founded the Nantes-based startup i-SEP around a novel blood auto-transfusion device that makes it possible to recover, treat, concentrate and re-administer blood from patients during surgery.
As Sylvain Picot explains: “Well, to begin with, this avoids risks due to incompatibility, contamination and post-operative complications induced by the transfusion of blood from a donor. Then we have other considerations such as supply-side blood shortages, the cost of purchasing from blood banks, and the medical and social impacts of side effects. In all, this new process can potentially generate millions in savings for hospitals. Our machine addresses issues related to Patient Blood Management, a field still relatively undeveloped in France. However, it is a real social issue for the patient and the healthcare system”.
Every year in France, more than one million patients receive a blood transfusion in the course of a surgical operation or during a emergency intervention. However, transfusion remains a delicate act given potential incompatibilities between donors and recipients.
From a commercial perspective, it is estimated that the auto-transfusion market is worth €300 million.
“More broadly, the potential is estimated at between 700 million to one billion euro” notes Sylvain Picot, whose solution is expected to deliver a blood-yield rate of 80% after filtration and adequate functionalities.
“In any case”, asserts Sylvain, [a rate] “well beyond competing devices”
Towards pre-commercialization in European in 2020
Thanks to partnerships engaged from the outset with the French National Blood Bank (EFS) and the Oniris Veterinary School in Nantes, i-SEP very quickly validated its hypotheses. In 2017, a consortium was created with the Bordeaux University Hospital and Oniris to apply for the PIA program in the PSPC category (Major Research and Development Projects supporting Competitiveness). Two partnerships were also concluded with the University Hospitals of Nantes and Rennes to extend the tests.
After conducting in-vitro tests on blood bags and in-vivo trials on animals, the validation process will continue during the second half of 2019 with clinical trials conducted over a period of four to six months on around 50 patients at three centers. The aim is to confirm the performance and safety of the device with a view to obtaining CE marking, thus clearing the way for the regulatory approvals required to launch the product on the market by 2020-2021.
At the same time, i-SEP, which holds fifteen international patents, has entered into discussions with the US Food and Drug Administration (FDA) to prepare the registration and export of the machine with a view to gaining a foothold in the transatlantic market. The latest prototype (consumable kit, pump, electronic board, touch screen, etc.) is currently being validated, following which it is planned to start considering mass production by 2021-2022. The first units, whose parts will be manufactured mainly in France, are expected to be assembled and tested early 2019 on-site in Nantes, where i-SEP currently employs twelve people. The commercial pre-launch is expected to take place in 2020 on pilot sites in Bordeaux, Rennes, Nantes, Lyon and on selected European centers in the United Kingdom and Germany.
Frédéric Thual